Physical mapping of 45S and 5S rDNA in two Sprekelia formosissima cytotypes (Amaryllidaceae) through Fluorescent In Situ Hybridization (FISH)
DOI:
https://doi.org/10.13128/caryologia-578Keywords:
bulbous genus, karyogram, karyotypic formula, ornamental, plant chromosomes, ploidy levelAbstract
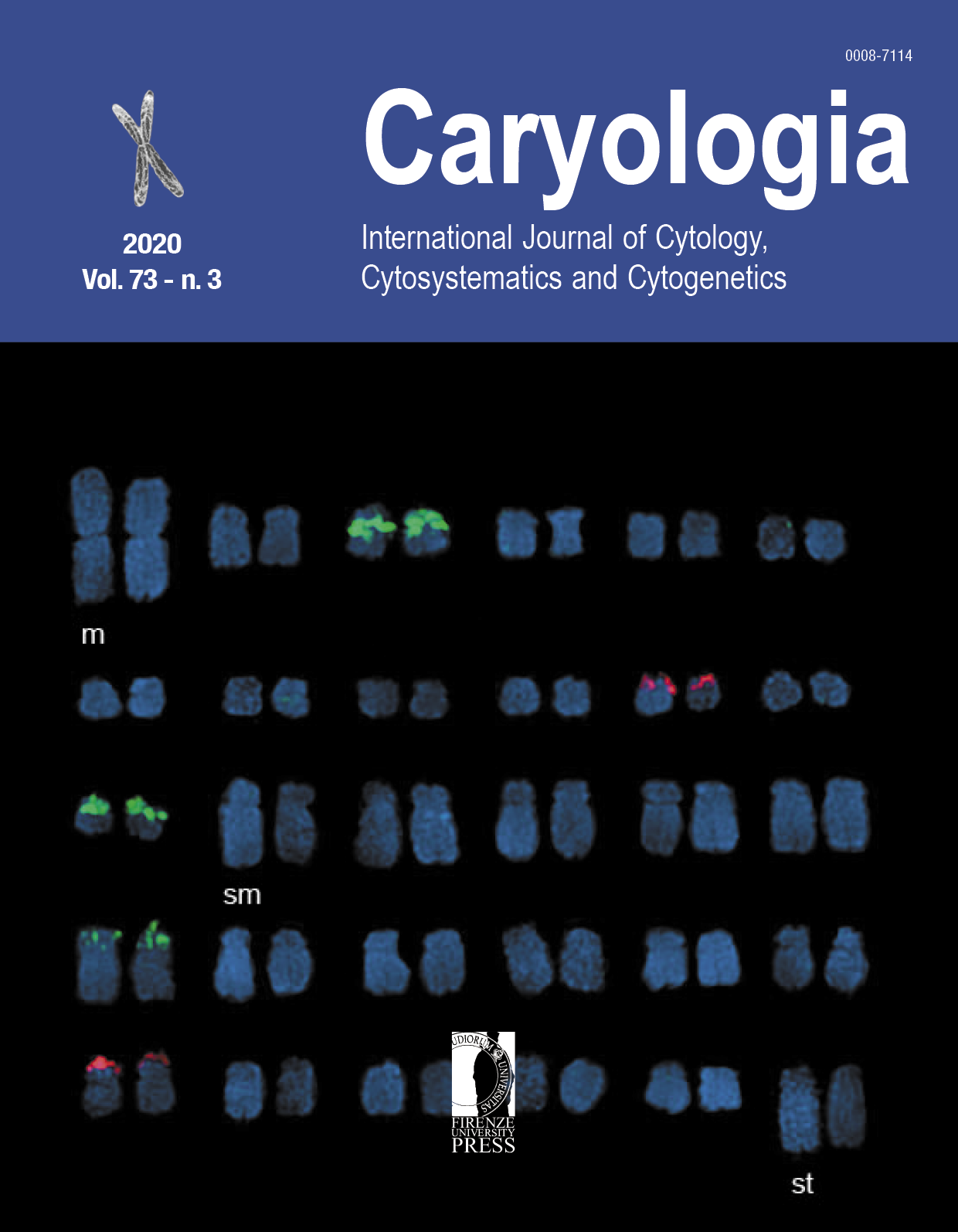
Chromosome number and position of rDNA were studied in plants of Sprekelia formosissima (Amaryllidaceae) collected in two populations with different ploidy level (2n=2x=60 and 2n=5x=150). The 5S and 45S rRNA loci were localized and physically mapped using two-color fluorescence in situ hybridization probes. The diploid (2n=2x=60) cytotype showed four loci for the 45S rDNA in two chromosome pairs (11 and 25) in telomeric position. The 5S rDNA was present in six loci of three homologous chromosome pairs (3, 13 and 19) in subtelomeric and telomeric positions. The chromosomes of the pentaploid cytotype (2n=5x=150) showed five loci for the 45S rDNA in telomeric position and five loci for the 5S rDNA in subtelomeric position. The karyotypic formula is 13m + 16sm + 1 st and the karyotype symmetry/asymmetry index is TF % = 34.67, AsK % = 65.32 and Syi % = 54.81, concluding that it is an asymmetric karyotype, bimodal with one distinctively large pair of chromosomes (10.42 µm) and a gradual decrease in the size of the other chromosome pairs, from the longest of 6.84 µm, to the shortest of 2.61 µm.
Downloads
References
Cabral JS, Felix LP, Guerra M. 2006. Heterochromatin diversity and its co-localization with 5S and 45S rDNA sites in chromosomes of four Maxillaria species (Orchidaceae). Genet Mol Biol. 29(4):659-664.
Clarkson JJ, Lim KY, Kova?ík A, Chase MW, Knapp S, Leitch AR. 2005. Long-term genome diploidization in allopolyploid Nicotiana section Repandae (Solanaceae). New Phytol. 168(1):241-252.
Deng CL, Qin RY, Wang NN, Cao Y, Gao J, Gao WJ, Lu LD. 2012. Karyotype of asparagus by physical mapping of 45S and 5S rDNA by FISH. J Genet. 91:209-212.
Ero?lu HE. 2015. Which chromosomes are subtelocentric or acrocentric? A new karyotype symmetry/asymmetry index. Caryologia. 68(3):239-245.
Flory WS. 1977. Overview of chromosomal evolution in the Amaryllidaceae. Nucleus. 20:70-88.
García BN. 2015. Systematics and evolution of Amaryllidaceae tribe Hippeastreae (Asparagales) [dissertation]. Gainesville (FL): University of Florida.
Gomez-Rodriguez VM, Rodriguez-Garay B, Palomino G, Martínez J, Barba-Gonzalez R. 2013. Physical mapping of 5S and 18S ribosomal DNA in three species of Agave (Asparagales, Asparagaceae). Comp Cytogenet. 7(3):191-203.
Gerlach WL, Bedbrook JR. 1979. Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucleic Acids Res. 7(7):1869-1885.
Gerlach WL, Dyer TA. 1980. Sequence organization of the repeating units in the nucleus of wheat which contain 5S rRNA genes. Nucleic Acids Res. 8(21):4851-4865.
Hwang YJ, Song CM, Younis A, Kim CK, Kang YI, Lim KB. 2015. Morphological characterization under different ecological habitats and physical mapping of 5S and 45S rDNA in Lilium distichum with fluorescence in situ hybridization. Rev Chil Hist Nat. 88(1):8.
Kotseruba V, Pistrick K, Blattner FR, Kumke K, Weiss O, Rutten T, Fuchs J, Endo T, Nasuda S, Ghukasyan A, Houben A. 2010. The evolution of the hexaploid grass Zingeria kochii (Mez) Tzvel. (2n=12) was accompanied by complex hybridization and uniparental loss of ribosomal DNA. Mol Phylogenet Evol. 56(1):146-155.
Kova?ík A, Pires JC, Leitch AR, Lim KY, Sherwood A, Matyášek R, Rocca J, Soltis DE, Soltis PS. 2005. Rapid concerted evolution of nuclear ribosomal DNA in two Tragopogon allopolyploids of recent and recurrent origin. Genetics. 169(2):931-944.
Levan A, Fredga K, Sandberg AA. 1964. Nomenclature for centromeric position on chromosomes. Hereditas. 52(2):201-220.
Li LJ, Arumuganathan K. 2001. Physical mapping of 45S and 5S rDNA on maize metaphase and sorted chromosomes by FISH. Hereditas. 134(2):141-145.
Lim K-B, Wennekes J, de Jong JH, Jacobsen E, Van Tuyl JM. 2001. Karyotype analysis of Lilium longiflorum and Lilium rubellum by chromosome banding and fluorescence in situ hybridisation. Genome. 44(5):911-918.
Liu B, Davis TM. 2011. Conservation and loss of ribosomal RNA gene sites in diploid and polyploid Fragaria (Rosaceae). BMC Plant Biol. 11(1):157.
López-Ferrari AR, Espejo-Serna A. 2002. Amaryllidaceae. In Sosa V, Rodríguez LC, Escamilla M, Moreno NP, Mejía-Saulés MT, Nee M, Nevling LI, Rzedowski J, editors. Flora de Veracruz Fasc. 128. México: Instituto de Ecología, A. C. and California (CA): University of California; p. 1-32.
Mizuochi H, Marasek A, Okazaki K. 2007. Molecular cloning of Tulipa fosteriana rDNA and subsequent FISH analysis yields cytogenetic organization of 5S rDNA and 45S rDNA in T. gesneriana and T. fosteriana. Euphytica. 155(1-2):235-248.
Mondin M, Santos-Serejo JA, Aguiar-Perecin ML. 2007. Karyotype characterization of Crotalaria juncea (L.) by chromosome banding and physical mapping of 18S-5.8 S-26S and 5S rRNA gene sites. Genet Mol Biol. 30(1):65-72.
Reeves A. 2001. MicroMeasure: a new computer program for the collection and analysis of cytogenetic data. Genome. 44(3):439-443.
Roa F, Guerra M. 2012. Distribution of 45S rDNA sites in chromosomes of plants: Structural and evolutionary implications. BMC Evol Biol. 12(1):225.
Robert ML, Lim KY, Hanson L, Sanchez-Teyer F, Bennett MD, Leitch AR, Leitch IJ. 2008. Wild and agronomically important Agave species (Asparagaceae) show proportional increases in chromosome number, genome size, and genetic markers with increasing ploidy. Bot J Linn Soc. 158(2):215-222.
Rodríguez-Domínguez JM, Ríos-Lara LL, Tapia-Campos E, Barba-Gonzalez R. 2017. An improved technique for obtaining well-spread metaphases from plants with numerous large chromosomes. Biotech Histochem. 92(3):159-166.
Sánchez SO. 1979. La Flora del Valle de México [The Flora of the Valley of Mexico]. México: Ed. Herrero, 5ª ed. p. 519.
Specht T, Szymanski M, Barciszewska MZ, Barciszewski J, Erdmann VA. 1997. Compilation of 5S rRNA and 5S rRNA gene sequences. Nucleic Acids Res. 25(1):96-97.
Stebbins GL. 1971. The Morphological, physiological and cytogenetic significance of polyploidy. In Barrington EJW, Willis AJ, editors. Chromosomal evolution in higher plants. London: Edward Arnold Ltd; p. 124-154.
Weiss-Schneeweiss H, Tremetsberger K, Schneeweiss GM, Parker JS, Stuessy TF. 2008. Karyotype diversi?cation and evolution in diploid and polyploid South American Hypochaeris (Asteraceae) inferred from rDNA localization and genetic ?ngerprint data. Ann Bot. 101(7):909-918.
Weiss-Schneeweiss H, Schneeweiss GM. 2013. Karyotype diversity and evolutionary trends in angiosperms. In Greilhuber J, Dolezel J, Wendel J, editors. Plant Genome Diversity Vol. 2. Vienna: Springer; p. 209-230.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2020 José Manuel Rodríguez Domínguez, Ernesto Tapia Campos, Rodrigo Barba Gonzalez

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Copyright on any open access article in a journal published byCaryologia is retained by the author(s).
- Authors grant Caryologia a license to publish the article and identify itself as the original publisher.
- Authors also grant any third party the right to use the article freely as long as its integrity is maintained and its original authors, citation details and publisher are identified.
- The Creative Commons Attribution License 4.0 formalizes these and other terms and conditions of publishing articles.
- In accordance with our Open Data policy, the Creative Commons CC0 1.0 Public Domain Dedication waiver applies to all published data in Caryologia open access articles.