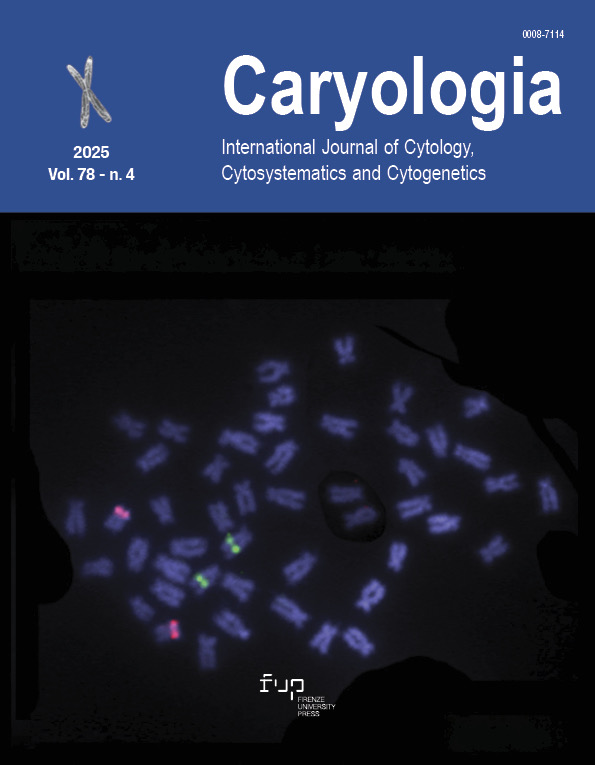
The genome organization of repetitive sequences in the Golden Damselfish, Amblyglyphidodon aureus (Cuvier, 1830) (Family Pomacentridae): insights from extensive pericentric inversions
DOI:
https://doi.org/10.36253/caryologia-3438Keywords:
Damselfishes, Chromosome, Repetitive sequences, repetitive DNAAbstract
The genus Amblyglyphidodon (damselfishes) is one of the most distinctive fish groups in the Indo-Pacific coral reef ecosystem. It consists of 11 recognized species, which display complex taxonomic relationships and cryptic lineages across their extensive range, often inhabiting coral environments. The timing of evolutionary divergences among these species varies greatly, and the cytogenetic events that accompanied their diversification remain poorly understood. In this regard, chromosomal divergence analysis from a phylogenetic viewpoint provides valuable insights into karyoevolutionary trends. In this study, we employed conventional cytogenetic and cytogenomic techniques to investigate the Golden Damselfish, Amblyglyphidodon aureus, focusing on mapping repetitive DNA elements and transposable elements, including 18S rDNA, 5S rDNA, (CA)15, (GA)15, and (CAA)10. The result show that A. aureus has a distinct karyotype (2n=48, FN=96), characterized by numerous bi-armed chromosomes, with pericentric inversions playing a key role in its karyotypic structure. Pericentric inversions often function as postzygotic reproductive barriers in many species, and this may also be the case for A. aureus. These chromosomal differences illustrate the contrasting evolutionary pathways within the Amblyglyphidodon genus, shedding light on the karyotypic characteristics that define the group.
Downloads
References
Allen GR, Erdmann MV, Cahyani DNK. Review of the Chrysipteraoxycephala complex of damselfishes (Pomacentridae) with descriptions of three new species from the East Indian Archipelago. Journal of the Ocean Science Foundation. 2015;17:56–84.
Allen GR, Erdmann MV, Purtiwi PD. Descriptions of four new species of damselfishes (Pomacentridae) in the Pomacentrus philippinus complex from the tropical western Pacific Ocean. Journal of the Ocean Science Foundation. 2017;25:47–76.
Allen GR, Randall JE. A review of the Leucogaster species complex of the Indo-pacific pomacentrid genus Amblyglyphidodon, with descriptions of two new species. aqua, International Journal of Ichthyology. 2002;5:139–152.
Allen GR. Conservation hotspots of biodiversity and endemism for Indo-Pacific coral reef fishes. Aquat Conser Mar Freshw Ecosyst. 2008;18:541–556. DOI: https://doi.org/10.1002/aqc.880
Arai R, Inoue M. Chromosomes of four species of coral fishes from Japan. Bulletin of the National Science Museum, Series A. 1976;2(2):137-141.
Arai R. Fish Karyotypes: a check list. Tokyo: Springer; 2011. DOI: https://doi.org/10.1007/978-4-431-53877-6
Belyayev A, Kalendar R, Brodsky L, Nevo E, Schulman AH, Raskina O. Transposable elements in a marginal plant population: temporal fluctuations provide new insights into genome evolution of wild diploid wheat. Mobile DNA. 2010; https://doi:10.1186/1759-8753-1-6. DOI: https://doi.org/10.1186/1759-8753-1-6
Bernardi G, Crane NL. Molecular phylogeny of the humbug damselfishes inferred from mtDNA sequences. J Fish Biol. 1999;54:1210–1217. DOI: https://doi.org/10.1111/j.1095-8649.1999.tb02049.x
Bertollo LAC, Cioffi MB, Moreira-Filho O: Direct chromosome preparation from freshwater teleost fishes. In: Ozouf-Costaz C, Pisano E, Foresti, F and Almeida Toledo LF, editors. Fish Cytogenetic Techniques (Chondrichthyans and Teleosts).CRC Press: Enfield, Boca Raton, FL; 2015. pp. 21–26.
Bone Q, Moore RH. Biology of fishes. 3rd ed. New York: Taylor & Francis Group, 2008. DOI: https://doi.org/10.1201/9781134186310
Coleman RR, Gaither MR, Kimokeo B, Stanton FG, Bowen BW, Toonen RJ. Large-scale introduction of the Indo-Pacific damselfish Abudefduf vaigiensis into Hawaii promotes genetic swamping of the endemic congener A. abdominalis. Mol Ecol. 2014;23:5552–5565. DOI: https://doi.org/10.1111/mec.12952
Cooper WJ, Smith LL, Westneat MW. Exploring the radiation of a diverse reef fish family: phylogenetics of the damselfishes (Pomacentridae), with new classifications based on molecular analyses of all genera. Molecular Phylogenetics and Evolution. 2009;52:1–16. DOI: https://doi.org/10.1016/j.ympev.2008.12.010
Cunha IMC, Souza AS, Dias EA, Amorim KDJ, Soares RX, Costa GWWF, García-Machado E, Galetti Jr PM, Molina WF. Genetic multipartitions based on d-loop sequences and chromosomal patterns in Brown Chromis, Chromis multilineata (Pomacentridae), in the Western Atlantic. BioMed Research International. 2014;254698. DOI: https://doi.org/10.1155/2014/254698
Dobigny G, Ozouf-Costaz C, Waters PD. LINE-1 amplification accompanies explosive genome repatterning in rodents. Chromosome Research. 2004;12:787. DOI: https://doi.org/10.1007/s10577-005-5265-y
Drew JA, Barber PH. Comparative phylogeography in Fijian coral reef fishes: a multi-taxa approach towards marine reserve design. PLoSOne. 2012; https://doi:10.1371/journal.pone.0047710. DOI: https://doi.org/10.1371/journal.pone.0047710
Eschmeyer WN, Fong JD. Title of subordinate document. In: Species by Family/Subfamily. 2016. http://researcharchive.cala cademy.org/research/ichthyology/catalog/SpeciesByFamily.asp. Accessed 15 Jan 2025.
Ferreira DC, Porto-Foresti F, Oliveira C, Foresti F. Transposable elements as a potential source for understanding the fish genome. Mobile Genetic Elements. 2011;1:2:112–117. DOI: https://doi.org/10.4161/mge.1.2.16731
Fishelson L. Behaviour, socio-ecology and sexuality in damselfishes (Pomacentridae). Italian Journal of Zoology. 1998;65:387–398. DOI: https://doi.org/10.1080/11250009809386853
Frédérich B, Olivier D, Litsios G, Alfaro ME, Parmentier E. Trait decoupling promotes evolutionary diversification of the trophic and acoustic system of damselfishes. Proc R Soc B. 2014;281:20141047. DOI: https://doi.org/10.1098/rspb.2014.1047
Frédérich B, Sorenson L, Santini F, Slater GJ, Alfaro ME. Iterative ecological radiation and convergence during the evolutionary history of damselfishes (Pomacentridae). Am Nat. 2013;181:94–113. DOI: https://doi.org/10.1086/668599
Fricke HW, Holzberg S. Social units and hermaphroditism in a pomacentrid fish. Naturwissensch. 1974;61:367–68. DOI: https://doi.org/10.1007/BF00600312
Gaither MR, Rocha LA. Origins of species richness in the Indo-Malay-Philippine biodiversity hotspot: evidence for the centre of overlap hypothesis Journal of Biogeography. 2013;40:1638–1648. DOI: https://doi.org/10.1111/jbi.12126
Galetti PM, Molina WF, Affonso PRAM. Assessing genetic diversity of Brazilian reef fishes by chromosomal and DNA markers. Genetica,. 2006;126(1):91–99. DOI: https://doi.org/10.1007/s10709-005-1446-z
Getlekha N, Cioffi MB, Yano CF, Maneechot N, Bertollo LAC, Supiwong W, Tanomtong A, Molina WF. Chromosome mapping of repetitive DNAs in sergeant major fishes (Abudefdufinae, Pomacentridae): a general view on the chromosomal conservatism of the genus. Genetica. 2016b;144:567–576. DOI: https://doi.org/10.1007/s10709-016-9925-y
Getlekha N, Molina WF, Cioffi MB, Yano CF, Maneechot N, Bertollo LAC, Supiwong W, Tanomtong A. Repetitive DNAs highlight the role of chromosomal fusions in the karyotype evolution of Dascyllus species (Pomacentridae, Perciformes). Genetica 2016a;144:203–211. DOI: https://doi.org/10.1007/s10709-016-9890-5
Getlekha, N, Cioffi MB, Maneechot N, Bertollo LAC, Supiwong W. Tanomtong A, Molina WF. Contrasting Evolutionary Paths Among Indo-Pacific Pomacentrus Species Promoted by Extensive Pericentric Inversions and Genome Organization of Repetitive Sequences. Zebrafish. 2018;15(1): 45–54. DOI: https://doi.org/10.1089/zeb.2017.1484
Hardie D C, Hebert PD. The nucleotypic effects of cellular DNA content in cartilaginous and ray-finned fishes. Genome. 2003; 46(5): 683–706. DOI: https://doi.org/10.1139/g03-040
Hardie DC, Hebert PD. Genome-size evolution in fishes. Can J Fish Aquatic Sci. 2004;61(9): 1636–1646. DOI: https://doi.org/10.1139/f04-106
Hoffmann AA, Rieseberg LH. Revisiting the impact of inversions in evolution: from population genetic markers to drivers of adaptive shifts and speciation? Annu Rev Ecol Evol Syst. 2008;39:21–42. DOI: https://doi.org/10.1146/annurev.ecolsys.39.110707.173532
Howell WM, Black DA. Controlled silver-staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method. Experientia. 1980;36:1014–1015. DOI: https://doi.org/10.1007/BF01953855
Jang-Liaw NH, Tang KL, Hui C-F, Shao K-T. Molecular phylogeny of 48 species of damselfishes (Perciformes: Pomacentridae) using 12S mtDNA sequences. Mol Phylogenet Evol. 2002;25:445–454. DOI: https://doi.org/10.1016/S1055-7903(02)00278-6
Johansen JL, He S, Frank G, Pappas MK, Berumen ML, Hoey AS. Hybridization between damselfishes Dascyllus aruanus and D. reticulatus on the Great Barrier Reef. Coral Reefs. 2017; https://doi:10.1007/s00338-017-1563-z. DOI: https://doi.org/10.1007/s00338-017-1563-z
Kashiwagi E, Takai A, Ojima Y. Chromosomal distribution of constitutive heterochromatin and nucleolus organizer regions in four Dascyllus fishes (Pomacentridae, Perciformes). Cytologia. 2005;70:345–349. DOI: https://doi.org/10.1508/cytologia.70.345
Kasiroek W, Luangoon N, Getlekha N, Saowakoon S, Phinrub W, Tanomtong A. First report on heteromorphic NORs and chromosome analysis of Rolland’s demoiselle, Chrysiptera rollandi (Perciformes, Pomacentrinae) by conventional and Ag-NOR staining techniques. Cytologia. 2014;79:289–297. DOI: https://doi.org/10.1508/cytologia.79.289
Kirkpatrick M. How and why chromosome inversions evolve. PLoS Biology, 2010; https://doi.org/10.1371/journal.pbio.1000501. DOI: https://doi.org/10.1371/journal.pbio.1000501
Kumar S, Blaxter M, Hebert PD. Complete mitochondrial genome of Staghorn damselfish, Amblyglyphidodon curacao (Perciformes: Pomacentridae). Mitochondrial DNA Part B. 2016; 1(1):1–2.
Lim JK, Simmons MJ. Gross chromosome rearrangements mediated by transposable elements in Drosophila melanogaster. Bioessays. 1994;16:269–275. DOI: https://doi.org/10.1002/bies.950160410
Litsios G, Pellissier L, Forest F, Lexer C, Pearman PB, Zimmermann NE, Salamin N, Trophic specialization influences the rate of environmental niche evolution in damselfishes (Pomacentridae). Proc Biol Sci. 2012;279:3662–3669. DOI: https://doi.org/10.1098/rspb.2012.1140
Liu SYV, Dai CF, Allen GR, Erdmann MV. Phylogeography of the neon damselfish Pomacentrus coelestis indicates a cryptic species and different species origins in the West Pacific Ocean. Mar Ecol Prog Ser. 2012;458:155–167. DOI: https://doi.org/10.3354/meps09648
Liu SYV, Ho HCH, Dai CF. A new species of Pomacentrus (Actinopterygii: Pomacentridae) from Micronesia, with comments on its phylogenetic relationships. Zool Stud. 2013;52:6–13. DOI: https://doi.org/10.1186/1810-522X-52-6
Marques DA, Lucek K, Meier JI, Mwaiko S, Wagner CE, Excoffier L, Seehausen O, Genomics of rapid incipient speciation in sympatric threespine stickleback. PLOS Genetics. 2016;12:2:e1005887. DOI: https://doi.org/10.1371/journal.pgen.1005887
Martins C, Ferreira IA, Oliveira C, Foresti F, Galetti Jr PM. A tandemly repetitive centromeric DNA sequence of the fish Hoplias malabaricus (Characiformes: Erythrinidae) is derived from 5S rDNA. Genetica. 2006;127:133–141. DOI: https://doi.org/10.1007/s10709-005-2674-y
Maruska KP, Peyton KA. Interspecific spawning between a recent immigrant and an endemic damselfish (Pisces: Pomacentridae) in the Hawaiian Islands. Pac Sci. 2007;61:211–221. DOI: https://doi.org/10.2984/1534-6188(2007)61[211:ISBARI]2.0.CO;2
Molina WF, Galetti Jr PM. Karyotypic changes associated to the dispersive potential on Pomacentridae (Pisces, Perciformes). J Exp Mar Biol Ecol. 2004a;309:109–119. DOI: https://doi.org/10.1016/j.jembe.2004.03.011
Molina WF, Galetti Jr PM. Multiple pericentric inversions and chromosomal divergence in the reef fishes Stegastes (Perciformes, Pomacentridae). Genet Mol Biol. 2004b;27:543–548. DOI: https://doi.org/10.1590/S1415-47572004000400013
Molina WF, Galetti Jr PM. Robertsonian rearrangements in the reef fish Chromis (Perciformes, Pomacentridae) involving chromosomes bearing 5S rRNA genes. Genet Mol Biol. 2002;25:373–377. DOI: https://doi.org/10.1590/S1415-47572002000400004
Molina WF, Martinez PA, Bertollo LA, Bidau CJ. Preferential accumulation of sex and Bs chromosomes in biarmed karyotypes by meiotic drive and rates of chromosomal changes in fishes. An Acad Bras Ciênc. 2014b;86:4:1801–1812. DOI: https://doi.org/10.1590/0001-3765201420130489
Molina WF. Chromosomal changes and stasis in marine fish groups. In: Pisano E, Ozouf-Costaz C, Foresti F, Kapoor BG. editors. Fish cytogenetics. Enfield: Science Publishers; 2007. pp. 69–110, DOI: https://doi.org/10.1201/b10746-4
Montanari SR, Hobbs JP, Pratchett MS, van Herwerden L. The importance of ecological and behavioural data in studies of hybridisation among marine fishes. Reviews in Fish Biology and Fisheries. 2016;26:181–198. DOI: https://doi.org/10.1007/s11160-016-9420-7
Nagpure NS, Kumar R, Srivastava SK, Kushwaha B, Gopalakrishnan A, Basheer VS. Cytogenetic characterization of two marine ornamental fishes, Chaetodon collare and Stegastes insularis. Journal of the Marine Biological Association of India. 2006; 48(2):267-269.
Naish T, Powell R, Levy R, Wilson G, Scherer R, Talarico F, Krissek L, Niessen F, Pompilio M, Wilson T. Obliquity-paced Pliocene West Antarctic ice sheet oscillations. Nature. 2009;458:322–328. DOI: https://doi.org/10.1038/nature07867
Ojima Y. Fish cytogenetics. In: Sharma AK, Sharma A, editors. Chromosomes in evolution of eukaryotic groups. CRC Press: Boca Raton; 1983.
Ojima Y. Yamamoto K. Cellular DNA contents of fishes determined by flow cytometry. La Kromosomo II. 1990;57:1871–1888.
Planes S, Doherty PJ, Bernardi G. Strong genetic divergence among populations of a marine fish with limited dispersal, Acanthochromis polyacanthus, within the Great Barrier reef and the Coral Sea. Evolution. 2001;55:2263–2273. DOI: https://doi.org/10.1111/j.0014-3820.2001.tb00741.x
Rishi KK. A preliminary report on the karyotypes of eighteen marine fishes. Research Bulletin of the Punjab University. 1973;24:161–162.
Rocha LA, Rocha CR, Robertson DR, Bowen BW. Comparative phylogeography of Atlantic reef fishes indicates both origin and accumulation of diversity in the Caribbean. BMC Evolutionary Biology. 2008; 8:157. DOI: https://doi.org/10.1186/1471-2148-8-157
Russell ST. Evolution of intrinsic post-zygotic reproductive isolation in fish. Ann Zool Fennici. 2003;40:321–329.
Sorenson L, Allen GR, Erdmann MV, Dai CF, Liu S-YV. Pleistocene diversification of the Pomacentrus coelestis species complex (Pisces: Pomacentridae): historical biogeography and species boundaries. Mar Biol. 2014;161:2495–2507. DOI: https://doi.org/10.1007/s00227-014-2521-8
Steinke D, Zemlak TS, Hebert PDN. Barcoding Nemo: DNA-based identifications for the ornamental fish trade. PLoS One 2009; 4:e6300. DOI: https://doi.org/10.1371/journal.pone.0006300
Sumner AT. A simple technique for demonstrating centromeric heterochromatin. Exp Cell Res. 1972;75:304–306. DOI: https://doi.org/10.1016/0014-4827(72)90558-7
Takai A, Ojima Y. Chromosome evolution associated with Robertsonian rearrangements in Pomacentrid fish (Perciformes). Cytobios. 1995;84:103–110.
Takai A, Ojima Y. Comparative studies of karyotypes and distributions of nucleolus organizer regions in pomacentrid fish. Cytobios. 1991;65:199–205.
Takai A, Ojima Y. Constitutive heterochromatin distribution in the chromosomes of Pomacentrid fishes (Perciformes). Cytologia. 1999;64:87–91. DOI: https://doi.org/10.1508/cytologia.64.87
Takai A. Chromosomal distribution of C-band-positive heterochromatin in eight species of Pomacentridae (Perciformes). Chromosome Science. 2012;15:39–42.
Takai A. Karyotype and nucleolar organizer regions in the pomacentrid fish Neoglyphidodon melas. Memoirs of Osaka Shin-Ai College. 2010; 45:9–13.
Takai A. Ojima Y. Comparative studies of karyotypes and distributions of nucleolus organizer regions in Pomacentrid fishes. Proceedings of the Japan Academy, Series B. 1986;62(1):17–22.
Takai A. Ojima Y. Comparative studies of karyotypes and distribution of nucleolus organizer regions in Pomacentrid fish. Proc Jpn Acad. 1987;63B: 17–20. DOI: https://doi.org/10.2183/pjab.63.17
Van Herwerden L, Doherty PJ. Contrasting genetic structures across two hybrid zones of a tropical reef fish, Acanthochromis polyacanthus (Bleeker 1855). Journal of Evolutionary Biology. 2006;19:239–252. DOI: https://doi.org/10.1111/j.1420-9101.2005.00969.x
Wellington GM, Victor BC. Planktonic larval duration of one hundred species of Pacific and Atlantic damselfishes (Pomacentridae). Marine Biology. 1989;101:557–567. DOI: https://doi.org/10.1007/BF00541659
Yano CF, Bertollo LAC, Cioffi MB. Fish-FISH: Molecular Cytogenetics in Fish Species. In: Liehr T, editors. Fluorescence in situ Hybridization (FISH)-Application Guide. 3rd ed. Springer: Berlin; 2017. pp. 429–444. DOI: https://doi.org/10.1007/978-3-662-52959-1_44
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Nuntaporn Getlekha, Kamika Sribenja

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Copyright on any open access article in a journal published byCaryologia is retained by the author(s).
- Authors grant Caryologia a license to publish the article and identify itself as the original publisher.
- Authors also grant any third party the right to use the article freely as long as its integrity is maintained and its original authors, citation details and publisher are identified.
- The Creative Commons Attribution License 4.0 formalizes these and other terms and conditions of publishing articles.
- In accordance with our Open Data policy, the Creative Commons CC0 1.0 Public Domain Dedication waiver applies to all published data in Caryologia open access articles.