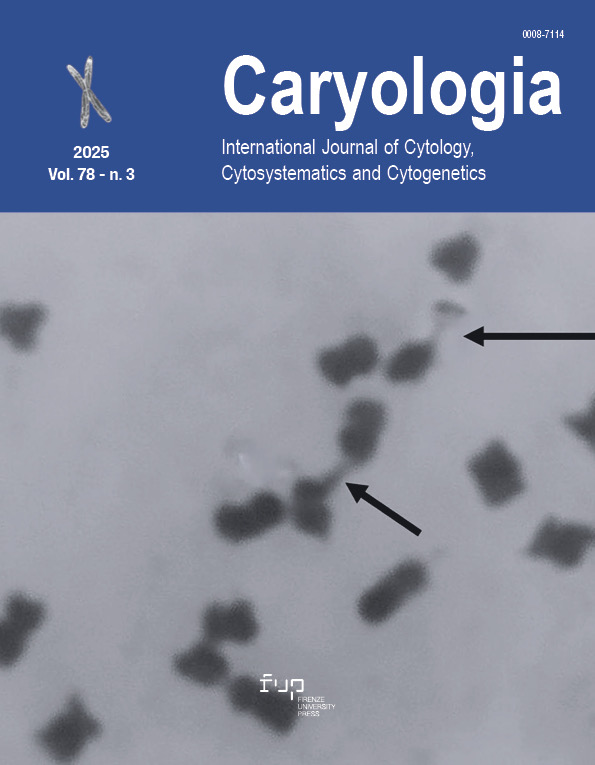
Spontaneous stickiness in somatic metaphase cells suggests chromosomal instability in a Mexican population of Aeschynomene sp. prope villosa (Fabaceae: Papilionoideae: Dalbergieae)
DOI:
https://doi.org/10.36253/caryologia-3492Keywords:
Chromosomal instability, chromosome stickiness, hybridization, interchromosomal connections, Karyotype, SAT-chromosomesAbstract
Aeschynomene sp. prope villosa (a dalbergioid legume) represents a group of populations with morphological characteristics similar to those of Aeschynomene villosa, but without complete overlap, and whose cytogenetic characteristics subtly differ from both the latter and other related taxa. In this study, conventional surface spread and air-drying techniques, along with Giemsa staining, were used to analyze metaphase chromosome complements and karyotypes of individuals belonging to a population of A. sp. prope villosa, a taxon included in the Americanae series of Aeschynomene. The results confirm a previously obtained karyotypic formula. Differences observed in chromosome sizes do not modify the karyotype or its symmetry. They are interpreted here as a loss of gene flow between populations or adaptations to different ecogeographic conditions. The presence of chromosome stickiness in 7.4% of the nuclei analyzed is noteworthy. This aberration, not previously observed in Aeschynomene, primarily involves areas near the telomeres of apparently homologous and non-homologous chromosomes. This phenomenon, observed mainly in meiosis, has been linked to the tendency of chromosomes to clump together during cellular divisions following plant hybridization events. The same criterion of chromosomal interaction after hybridization could explain the changes in the number and position of satellites recorded in a previous study. Although unexpected, these results should not be surprising, since intergradation between A. villosa and A. americana, species with which it overlaps in its distribution area, has been suspected for at least 70 years. Furthermore, hybridization and allopolyploidy have already been demonstrated in other morphological series of Aeschynomene.
Downloads
References
Alazar D, Becker M. 1987. Aeschynomene as green manure for rice. Plant Soil 101:141-143. https://doi.org/10.1007/BF02371043 DOI: https://doi.org/10.1007/BF02371043
Al-Achkar W, Sabatier L, Dutrillaux B. 1989. How are sticky chromosomes formed? Ann Genet. 32:10-15.
Arbo MM. 1985. Notas taxonómicas sobre Turneráceas Sudamericanas. Candollea 40(1):175-191.
Arrighi JF, Chaintreuil C, Cartieaux F, Cardi C, Rodier-Goud M, Brown SC, Boursot M, D´Hont A, Dreyfus B, Giraud E. 2014. Radiation of the Nod-independent Aeschynomene relies on multiple allopolyploid speciation events. New Phytol. 201(4):1457-1468. https://doi.org/10.1111/nph.12594 DOI: https://doi.org/10.1111/nph.12594
Beadle GW. 1932. A gene in Zea mays for failure of cytokinesis during meiosis. Cytologia 3(2):142-155. https://doi.org/10.1508/cytologia.3.142 DOI: https://doi.org/10.1508/cytologia.3.142
Brottier L, Chaintreuil C, Simion P, Scornavacca C, Rivallan R, Mournet P, Moulin L, Lewis GP, Fardoux J, Brown SC, et al. 2018. A phylogenetic framework of the legume genus Aeschynomene for comparative genetic analysis of the Nod-dependent and Nod-independent symbioses. BMC Plant Biol. 18:333. https://doi.org/10.1186/s12870-018-1567-z DOI: https://doi.org/10.1186/s12870-018-1567-z
Cardoso D, de Queiroz LP, Pennington RT, de Lima HC, Fonty E, Wojciechowski MF, Lavin M. 2012. Revisiting the phylogeny of papilionoid legumes: New insights from comprehensively sampled early-branching lineages. Am J Bot. 99(12):1991-2013. https://doi.org/10.3732/ajb.1200380 DOI: https://doi.org/10.3732/ajb.1200380
Cardoso DBOS, Mattos CMJ, Filardi F, Delgado-Salinas A, Lavin M, Moraes PLR, Tapia-Pastrana F, Lima HC. 2020. A molecular phylogeny of the pantropical papilionoid legume Aeschynomene supports reinstating the ecologically and morphologically coherent genus Ctenodon. Neodiversity 13:1-38. https://doi.org/10.13102/neod.131.1 DOI: https://doi.org/10.13102/neod.131.1
Chaintreuil C, Arrighi JF, Giraud E, Miché L, Moulin L, Dreyfus B, Munive-Hernández JA, Villegas-Hernández MC, Béna G. 2013. Evolution of symbiosis in the legume genus Aeschynomene. New Phytol. 200(4):1247-1259. https://doi.org/10.1111/nph.12424 DOI: https://doi.org/10.1111/nph.12424
Chauhan KPS, Abel WO. 1968. Evidence for the association of homologous chromosomes during premeiotic stages in Impatiens and Salvia. Chromosoma 25:297-302. https://doi.org/10.1007/BF01183122 DOI: https://doi.org/10.1007/BF01183122
Dowd MA, Gaulden ME, Proctor BL, Seibert GB. 1986. Formaldehyde-induced acentric chromosome fragments and chromosome stickiness in Chortophaga neuroblasts. Environ Mutagen. 8:401-411. https://doi.org/10.1002/em.2860080309 DOI: https://doi.org/10.1002/em.2860080309
Evans HJ. 1962. Chromosome aberrations induced by ionizing radiations. Int Rev Cytol. 13:221-321. https://doi.org/10.1016/S0074-7696(08)60285-5 DOI: https://doi.org/10.1016/S0074-7696(08)60285-5
Fernandes A. 1996. O táxon Aeschynomene no Brasil. Fortaleza: Edições UFC, Brasil, 130 pp.
Fontdevila A. 1992. Genetic instability and rapid speciation: are they coupled? Genetica 86(1-3):247-258. https://doi.org/10.1007/BF00133723 DOI: https://doi.org/10.1007/BF00133723
Fontdevila A. 2005. Hybrid genome evolution by transposition. Cytogenet Genome Res. 110:49-55. https://doi.org/10.1159/000084937 DOI: https://doi.org/10.1159/000084937
Gaulden ME. 1987. Hypothesis: some mutagens directly alter specific chromosomal proteins (DNA topoisomerase II and peripheral proteins) to produce chromosome stickiness, which causes chromosome aberrations. Mutagenesis 2:357-365. https://doi.org/10.1093/mutage/2.5.357 DOI: https://doi.org/10.1093/mutage/2.5.357
Golubovskaya IN. 1989. Meiosis in maize: mei-genes and conception of genetic control of meiosis. Adv Genet. 26:149-192. https://doi.org/10.1016/S0065-2660(08)60225-4 DOI: https://doi.org/10.1016/S0065-2660(08)60225-4
Grant WF. 1978. Chromosome aberrations in plants as a monitoring system. Environ Health Perspect. 27:37-43. https://doi.org/10.1289/ehp.782737 DOI: https://doi.org/10.1289/ehp.782737
Hiraoka T. 1958. Somatic syndesis in Daphne odora. I. The chromosome behaviour in mitosis. Proc Jap Acad. 34:700-705. DOI: https://doi.org/10.2183/pjab1945.34.700
Huziwara Y. 1962. Karyotype analysis in some genera of Compositae. VIII. Further studies on the chromosomes of Aster. Am J Bot. 49:116-119. https://doi.org/10.1002/j.1537-2197.1962.tb14916.x DOI: https://doi.org/10.1002/j.1537-2197.1962.tb14916.x
Kaur D, Singhal VK. 2019. Meiotic abnormalities affect genetic constitution and pollen viability in dicots from Indian cold deserts. BMC Plant Biol. 19:10. https://doi.org/10.1186/s12870-018-1596-7 DOI: https://doi.org/10.1186/s12870-018-1596-7
Kiihl PRP, Pereira ARA, Godoy SMD, Stenzel NMC, Risso-Pascotto C. 2011. Chromosome stickiness during meiotic behavior analysis of Passiflora serrato-digitata L. (Passifloraceae). Cienc Rural 41:1018-1023. http://www.redalyc.org/articulo.oa?id=33119153028 DOI: https://doi.org/10.1590/S0103-84782011005000076
Klášterská I, Natarajan AT. 1975. Stickiness in Rosa meiosis induced by hybridisation. Caryologia 28:81-88. https://doi.org/10.1080/00087114.1975.10796599 DOI: https://doi.org/10.1080/00087114.1975.10796599
Klášterská I, Natarajan AT, Ramel C. 1976. An interpretation of the origin of subchromatid aberrations and chromosome stickiness as a category of chromatid aberrations. Hereditas 83(2):153-162. https://doi.org/10.1111/j.1601-5223.1976.tb01581.x DOI: https://doi.org/10.1111/j.1601-5223.1976.tb01581.x
Lavin M, Pennington RT, Klitgaard BB, Sprent JI, de Lima HC, Gasson PE. 2001. The dalbergioid legumes (Fabaceae): delimitation of a pantropical monophyletic clade. Am J Bot. 88(3):503-533. https://doi.org/10.2307/2657116 DOI: https://doi.org/10.2307/2657116
Levan A, Fredga K, Sandberg AA. 1964. Nomenclature for centromeric position on chromosomes. Hereditas 52:201-219. http://dx.doi.org/10.1111/j.1601-5223.1964.tb01953.x DOI: https://doi.org/10.1111/j.1601-5223.1964.tb01953.x
Loidl J, Greilhuber J. 1983. Structural changes of Ag-stained nucleolus organizing regions and nucleoli during meiosis in Allium flavum. Can J Genet Cytol. 25(5):524-529. https://doi.org/10.1139/g83-079 DOI: https://doi.org/10.1139/g83-079
McGill M, Pathak S, Hsu TC. 1974. Effects of ethidium bromide on mitosis and chromosomes: A possible material basis for chromosome stickiness. Chromosoma 47:157-166. https://doi.org/10.1007/BF00331803 DOI: https://doi.org/10.1007/BF00331803
McVaugh R. 1987. Flora Novo-Galiciana (Leguminosae) V. University of Michigan Press. Ann Arbor, EUA. 786 pp.
Mehra RC, Rai KS. 1970. Cytogenetic studies of meiotic abnormalities in Collinsia tinctoria. I. Chromosomal stickiness. Can J Genet Cytol. 12:560-569. https://doi.org/10.1139/g70-075 DOI: https://doi.org/10.1139/g70-075
Mendes-Bonato AB, Pagliarini MS, Do Valle CB, Penteado MIDO. 2001. A severe case of chromosome stickiness in pollen mother cells of Brachiaria brizantha (Hochst.) Stapf (Gramineae). Cytologia 66(3):287-291. http://dx.doi.org/10.1508/cytologia.66.287 DOI: https://doi.org/10.1508/cytologia.66.287
Metcalfe CJ, Bulazel KV, Ferreri GC, Schroeder-Reiter E, Wanner G, Rens W, Obergfell C, Eldridge MDB, O’Neill RJ. 2007. Genomic instability within centromeres of interspecific marsupial hybrids. Genetics 177(4):2507-2515. https://doi.org/10.1534/genetics.107.082313 DOI: https://doi.org/10.1534/genetics.107.082313
Mitra J, Steward FC. 1961. Growth induction in cultures of Haplopappus gracilis. II. The behaviour of the nucleus. Amer J Bot. 48(5):358-368. https://doi.org/10.2307/2439327 DOI: https://doi.org/10.1002/j.1537-2197.1961.tb11651.x
Moav R. 1961. Genetic instability in Nicotiana hybrids. II. Studies of the Ws (pbg) locus of N. plumbaginifolia in N. tabacum nuclei. Genetics 46(9):1069-1087. https://doi.org/10.1093/genetics/46.9.1069 DOI: https://doi.org/10.1093/genetics/46.9.1069
Moav J, Moav R, Zohary D. 1968. Spontaneous morphological alterations of chromosomes in Nicotiana hybrids. Genetics 59(1):57-63. https://doi.org/10.1093/genetics/59.1.57 DOI: https://doi.org/10.1093/genetics/59.1.57
Olvera-Luna AR, Gama-López S, Delgado-Salinas A. 2012. Flora del Valle de Tehuacán-Cuicatlán: Fabaceae tribu Aeschynomeneae. Universidad Nacional Autónoma de México, Instituto de Biología.
Pathak S, McGill M, Hsu TC. 1975. Actinomycin D effects on mitosis and chromosomes: sticky chromatids and localized lesions. Chromosoma 50:79-88. https://doi.org/10.1007/bf00284964 DOI: https://doi.org/10.1007/BF00284964
Pessim C, Pagliarini MS, Silva N, Jank L. 2015. Chromosome stickiness impairs meiosis and influences reproductive success in Panicum maximum (Poaceae) hybrid plants. Genet Mol Res. 14(2):4195-4202. https://doi.org/10.4238/2015.April.28.2 DOI: https://doi.org/10.4238/2015.April.28.2
Pich U, Fuchs J, Schubert I. 1996. How do Alliaceae stabilize their chromosome ends in the absence of TTTAGGG sequences? Chromosome Res. 4(3):207-213. https://doi.org/10.1007/BF02254961 DOI: https://doi.org/10.1007/BF02254961
Puizina J, Siroky J, Mokros P, Schweizer D, Riha K. 2004. Mre11 deficiency in Arabidopsis is associated with chromosomal instability in somatic cells and Spo11-dependent genome fragmentation during meiosis. Plant Cell 16:1968-1978. https://doi.org/10.1105/tpc.104.022749 DOI: https://doi.org/10.1105/tpc.104.022749
Rao PN, Ranganadham P, Nirmala A. 1990. Behaviour of a sticky desynaptic mutant in pearl millet. Genetica 81:221-227. https://doi.org/10.1007/BF00360869 DOI: https://doi.org/10.1007/BF00360869
Reynolds ST. 1990. Aeschynomeneae (Benth.) Hutch. (Leguminosae) in Australia. Austrobaileya 3(2):177-202. https://www.jstor.org/stable/41738754 DOI: https://doi.org/10.5962/p.365742
Rodríguez G. 1990. El género Aeschynomeme L. (Leguminosae: Papilionoideae) en Venezuela. Sinopsis. Acta Bot Venez. 16(1):117-136. https://www.jstor.org/stable/41740492
Rudd VE. 1955. The American species of Aeschynomene. Contr US Natl Herb. 32:1-172.
Sato S. 1981. Cytological studies on the satellited chromosomes of Allium cepa. Caryologia 34(4):431-440. https://doi.org/10.1080/00087114.1981.10796911 DOI: https://doi.org/10.1080/00087114.1981.10796911
Schubert I. 1984. Mobile nucleolus organizing regions (NORs) in Allium (Liliaceae s. lat)? Inferences from the specifity of silver staining. Plant Syst Evol. 144(3-4):291-305. https://doi.org/10.1007/BF00984139 DOI: https://doi.org/10.1007/BF00984139
Schubert I, Ohle H, Hanelt P. 1983. Phylogenetic conclusions from Giemsa banding and NOR staining in Top Onions (Liliaceae). Plant Syst Evol. 143(4):245-256. https://doi.org/10.1007/BF00986607 DOI: https://doi.org/10.1007/BF00986607
Schubert I, Wobus U. 1985. In situ hybridization confirms jumping nucleolus organizing regions in Allium. Chromosoma 92(2):143-148. https://doi.org/10.1007/BF00328466 DOI: https://doi.org/10.1007/BF00328466
Sheidai M, Noormohamadi Z, Mirabdolbaghi-Kashani N, Ahmadi MR. 2003. Cytogenetic study of some rapeseed (Brassica napus L.) cultivars and their hybrids. Caryologia 56(4):387-397. https://doi.org/10.1080/00087114.2003.10589349 DOI: https://doi.org/10.1080/00087114.2003.10589349
Solís Neffa VG, Fernández A. 2002. Karyotypic studies in Turnera sidoides complex (Turneraceae, Leiocarpae). Am. J Bot. 89(4):551-558. https://doi.org/10.3732/ajb.89.4.551 DOI: https://doi.org/10.3732/ajb.89.4.551
Sosnikhina SP. 1973. Genetic control of the behaviour of chromosomes in meiosis in inbred lines of diploid rye (Secale cereale L.) 2. Abnormalities on the stages of Anaphase I and II tetrads. Genetika 9:21-26.
Souza MC, Vianna LF, Kawakita K, Miotto STS. 2012. O genero Aeschynomene L. (Leguminosae, Faboideae, Dalbergieae) na planicie de inundacao do alto rio Parana, Brasil. Braz. J. Biol. 10:198-210.
Tapia-Pastrana F, Delgado-Salinas A. 2020. First cytogenetic register of an allopolyploid lineage of the genus Aeschynomene (Leguminosae, Papilionoideae) native to Mexico. Caryologia 73(4):17-26. https://doi.org/10.13128/caryologia-949 DOI: https://doi.org/10.13128/caryologia-949
Tapia-Pastrana F, Delgado-Salinas A, Caballero J. 2020. Patterns of chromosomal variation in Mexican species of Aeschynomene (Fabaceae, Papilionoideae) and their evolutionary and taxonomic implications. Comp Cytogenet. 14:157-182. https://doi.org/10.3897/CompCytogen.v14i1.47264 DOI: https://doi.org/10.3897/CompCytogen.v14i1.47264
Tapia-Pastrana F, Mercado-Ruaro P. 2001. A combination of the “squash” and “splash” techniques to obtain the karyotype and asses meiotic behavior of Prosopis laevigata L. (Fabaceae: Mimosoideae). Cytologia 66:11-17. https://doi.org/10.1508/cytologia.66.11 DOI: https://doi.org/10.1508/cytologia.66.11
Therman E. 1951. Somatic and secondary pairing in Ornithogalum. Heredity 5:253-269. DOI: https://doi.org/10.1038/hdy.1951.23
Wagenaar EB. 1969. End-to-end chromosome attachments in mitotic interphase and their possible significance to meiotic chromosome pairing. Chromosoma 26:410-426. https://doi.org/10.1007/BF00326353 DOI: https://doi.org/10.1007/BF00326353
WFO (The World Flora Online). Aeschynomene villosa var. villosa. Published on the internet http://www.worldfloraonline.org/taxon/wfo-0000179554. [accessed on: 21 Feb 2025].
Wojciechowski MF, Lavin M, Sanderson MJ. 2004. A phylogeny of legumes (Leguminosae) based on analysis of the plastid matK gene resolves many well-supported subclades within the family. Am J Bot. 91(11):1846-1862. https://doi.org/10.3732/ajb.91.11.1846 DOI: https://doi.org/10.3732/ajb.91.11.1846
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Fernando Tapia-Pastrana

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Copyright on any open access article in a journal published byCaryologia is retained by the author(s).
- Authors grant Caryologia a license to publish the article and identify itself as the original publisher.
- Authors also grant any third party the right to use the article freely as long as its integrity is maintained and its original authors, citation details and publisher are identified.
- The Creative Commons Attribution License 4.0 formalizes these and other terms and conditions of publishing articles.
- In accordance with our Open Data policy, the Creative Commons CC0 1.0 Public Domain Dedication waiver applies to all published data in Caryologia open access articles.