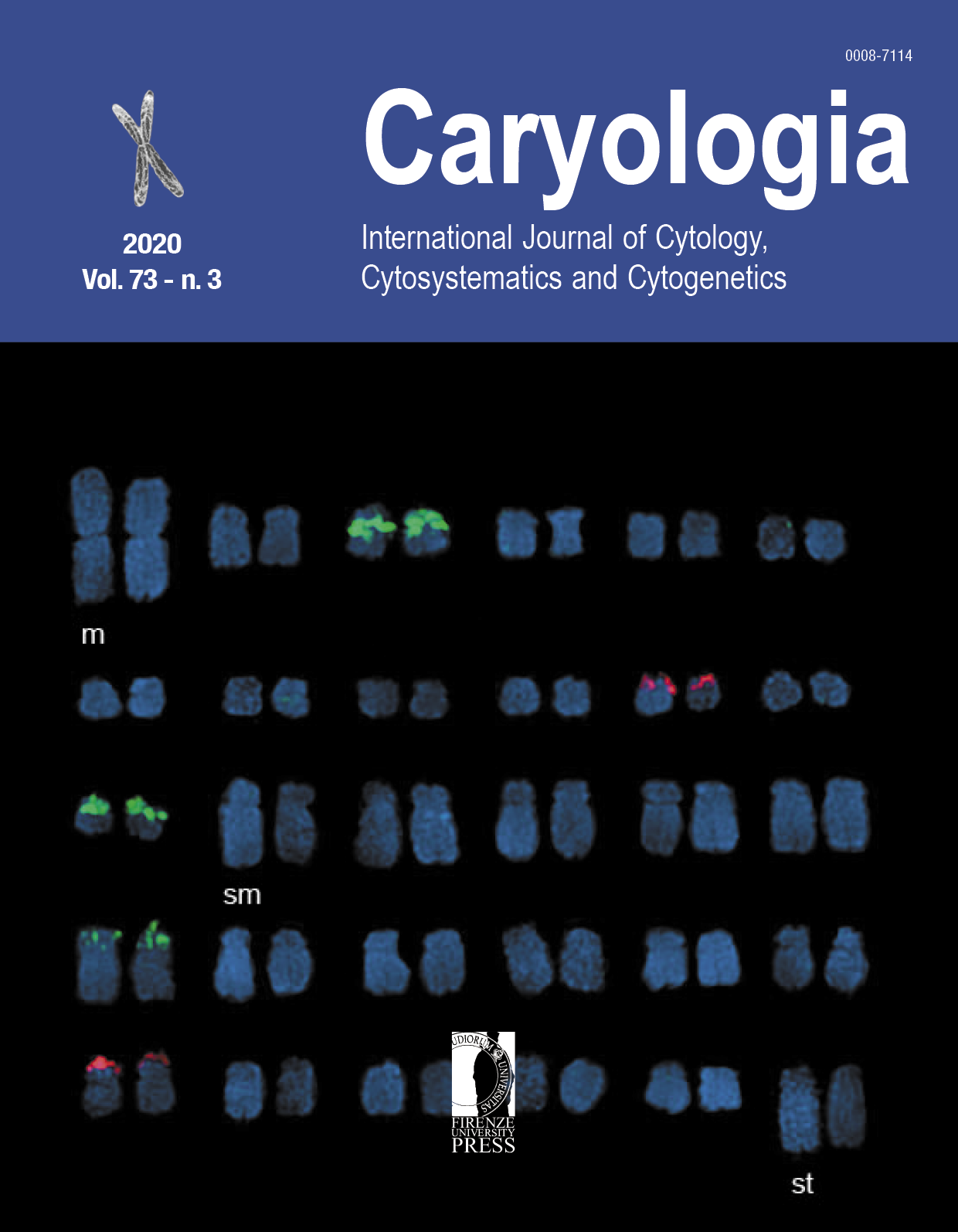
Display of Sukkula distributions on Barley Roots via in situ hybridization
DOI:
https://doi.org/10.13128/caryologia-191Keywords:
fluorescence in situ hybridization, retrotransposon, Sukkula, Barley, LTRs, internal-domainAbstract
Retrotransposon are an abundant and ancient parts of the plant genomes that especially LTR retrotransposons influence the genome size and evolution. Sukkula is a non-autonomous and active, relatively high copy-number retroelement. In this study, we performed fluorescence in situ hybridization (FISH) to observe the distributions of Sukkula elements (LTRs and internal-domain) by using labelled-PCR products. The localizations of Sukkula elements (LTRs and internal-domain) were observed under confocal microscope on Hordeum vulgare L. cv. Hasat root preparations. Our results revealed that Sukkula elements is still active and spread through the whole barley chromosomes. Additionally, the re-sequencing analysis of Sukkula LTRs demonstrated that LTRs sequences had ~65 bp gain. These analyses represent a valuable resource to reveal genome organization of barley and large sized plants.
Downloads
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. 1990. Basic local alignment search tool. J Mol Biol. 215:403-410.
Bennett MD, Leitch IJ. 2005. Nuclear DNA amounts in angiosperms: progress, problems and prospects. Ann Bot. 95:45–90.
Bustamante FO, Aliyeva-Schnorr L, Fuchs J, Beier S, Houben A. 2017. Correlating the genetic and physical map of barley chromosome 3H revealed limitations of the FISH-based mapping of nearby single-copy probes caused by the dynamic structure of metaphase chromosomes. Cytogenet Genome Res. 152:90–96.
Doolittle WF, Sapienza C. 1980. Selfish genes, the phenotype paradigm and genome evolution. Nature. 284:601–603.
Eickbush TH, Malik HS. 2002. Origin and evolution of retrotransposons. In: Craig NL, Craigie R, Gellert M, Lambowitz AM, editors. Mobile DNA. Washington (DC): ASM Press; p. 1111–1146.
Finnegan DJ. 1989. Eukaryotic transposable elements and genome evolution. Trends Genet. 5:103–107.
Flavell RB. 1986. Repetitive DNA and chromosome evolution in plants. Phil Trans R Soc London B: Biol Sci. 312:227–42.
Hausmann M, Cremer C. 2003. Standardisation of FISH-procedures: Summary of the first discussion workshop. Anal Cell Pathol. 25:201–205.
Havecker ER, Gao X, Voytas DF. 2004. The diversity of LTR retrotransposons. Genome Biol. 5:225
Hawkins JS, Kim H, Nason JD, Wing RA, Wendel JF. 2006. Differential lineage-specific amplification of transposable elements is responsible for genome size variation in Gossypium. Genome Res. 16:1252–1261.
Jenkins G, Hasterok R. 2001. Painting whole chromosome sets in hybrids using GISH. In: Advanced Molecular Cytogenetics – a practical course manual. Wydawnictwo Uniwersytetu ?l?skiego. p. 35-48.
Jenkins G, Hasterok R. 2007. BAC 'landing' on chromosomes of Brachypodium distachyon for comparative genome alignment. Nat Protoc. 2:88-98.
Kalendar R, Vicient CM, Peleg O, Anamthawat-Jonsson K, Bolshoy A, Schulman AH. 2004. Large Retrotransposon Derivatives: Abundant, Conserved but Nonautonomous Retroelements of Barley and Related Genomes. Genetics. 166(3):1437-1450.
Karlik E, Marakli S, Gozukirmizi N. 2018. Two lncRNAs expression profiles in salt stressed barley (Hordeum vulgare L.) roots. Cytologia. 83:37–43.
Kartal-Alacam G, Y?lmaz S, Marakli S, Gozukirmizi N. 2014. Sukkula Retrotransposon Insertion Polymorphism in Barley. Russ J Plant Physiol. 61(6):8282-833.
Kejnovsky E, Hawkins JS, Feschotte C. 2012. Plant transposable elements: biology and evolution. In: Wendel J, Greilhuber J, Dolezel J, Leitch IJ, editors. Plant Genome Diversity. Vol 1. India: Springer; p. 17-34.
Kemekawa N, Ohtsubo H, Horiuchi T, Ohtsubo E. 1999. Identification and characterization of novel retrotransposons of the gypsy type in rice. Mol Gen Genet. 260:593–602.
Kiseleva AV, Kirov IV, Khrustaleva LI. 2014. Chromosomal organization of centromeric Ty3/gypsy retrotransposons in Allium cepa L. and Allium fistulosum L. Genetika. 50:670–676.
Li Y, Zuo S, Zhang Z, Li Z, Han J, Chu Z, Hasterok R, Wang K. 2018. Centromeric DNA characterization in the model grass Brachypodium distachyon provides insights on the evolution of the genus. Plant J. 93(6):1088-1101.
Lin JY, Jacobus BH, SanMiguel P, Walling JG, Yuan Y, Shoemaker RC, Young ND, Jackson SA. 2005. Pericentromeric regions of soybean (Glycine max L. Merr.) chromosomes consist of retroelements and tandemly repeated DNA and are structurally and evolutionarily labile. Genetics. 170:1221–1230.
Lisch D. 2013. How important are transposons for plant evolution?. Nat Rev Genet. 14:49–61.
Ma J, Devos KM, Bennetzen JL. 2004. Analyses of LTR retrotransposon structures reveal recent and rapid genomic DNA loss in rice. Genome Res. 14:860–869.
Mafra I, Silva SA, Moreira EJMO, Ferreira da Silva CS, Beatriz M, Oliveira PP. 2008. Comparative study of DNA extraction methods for soybean derived food products. Food Control. 19:1183–1190.
Manninen I, Schulman AH. 1993. BARE-1, a copia-like retroelemnt in barley (Hordeum vulgare L.). Plant Mol Biol. 22:829–846.
Mascher M, Gundlach H, Himmelbach A, Beier S, Twardziok SO, Wicker T, Radchuk V, Dockter C, Hedley PE, Russell J, et al. 2017. A chromosome conformation capture ordered sequence of the barley genome. Nature. 544(7651):427-433.
Neumann P, Kobl?´zkova´ A, Navra´tilova´ A, Macas J. 2006. Significant expansion of Vicia pannonica genome size mediated by amplification of a single type of giant retroelement. Genetics. 173:1047–1056.
Orgel LE, Crick FHC. 1980. Selfish DNA: the ultimate parasite. Nature. 284:604–607.
Rebollo R, Romanish MT, Mager DL. 2012. Transposable elements: an abundant and natural source of regulatory sequences for host genes. Annu Rev Genet. 46:21–42.
Salvo-Garrido HG, Travella S, Schwarzacher T, Harwood WA, Snape JW. 2001. An efficient method for the physical mapping of transgenes in barley using in situ hybridization. Genome. 44:104–110.
SanMiguel P, Tikhonov A, Jin YK, Motchoulskaia N, Zakharov D, Melake-Berhan A, Springer PS, Edwards KJ, Lee M, Avramova Z, et al. 1996. Nested retrotransposons in the intergenic regions of the maize genome. Science. 274:765–768.
Schnable PS, Ware D, Fulton RS, Stein JC, Wei F, Pasternak S, Liang C, Zhang J, Fulton L, Graves TA, et al. 2009. The B73 maize genome: complexity, diversity, and dynamics. Science. 326:1112–1115.
Schulman AH, and Kalendar R. 2005. A movable feast: diverse retrotransposons and their contribution to barley genome Dynamics. Cytogenet Genome Res. 110:598-605.
Shams I, Raskina O. 2018. Intraspecific and intraorganismal copy number dynamics of retrotransposons and tandem repeat in Aegilops speltoides Tausch (Poaceae, Triticeae). Protoplasma. 255:1023–1038.
Shirasu K, Schulman AH, Lahaye T, Schulze-Lefert P. 2000 A contiguous 66 kb barley DNA sequence provides evidence for reversible genome expansion. Genome Res. 10:908–915.
Stephens JL, Brown SE, Lapitan NLV, Knudson DL. 2004. Physical mapping of barley genes using an ultrasensitive fluorescence in situ hybridization technique. Genome. 47:179–189.
Vicient CM, Suoniemi A, Anamthawat-Jo´nsson K, Tanskanen J, Beharav A, Nevo E, Schulman AH. 1999. Retrotransposon BARE-1 and its role in genome evolution in the genus hordeum. Plant Cell. 11:1769–1784.
Vitte C, Bennetzen JL. 2006 Analysis of retrotransposon structural diversity uncovers properties and propensities in angiosperm genome evolution. Proc Natl Acad Sci USA. 103:17638–17643.
Wessler SR, Bureau TE, White SE. 1995. LTRretrotransposons and MITEs: important players in the evolution of plant genomes. Curr Opin Genet Dev. 5(6):814-821.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2020 Elif Karlik

This work is licensed under a Creative Commons Attribution 4.0 International License.
- Copyright on any open access article in a journal published byCaryologia is retained by the author(s).
- Authors grant Caryologia a license to publish the article and identify itself as the original publisher.
- Authors also grant any third party the right to use the article freely as long as its integrity is maintained and its original authors, citation details and publisher are identified.
- The Creative Commons Attribution License 4.0 formalizes these and other terms and conditions of publishing articles.
- In accordance with our Open Data policy, the Creative Commons CC0 1.0 Public Domain Dedication waiver applies to all published data in Caryologia open access articles.